The bond angles for the molecules having a tetrahedral geometry is 109 but as the geometry of H2O molecule is distorted due to the presence of the lone pairs of electrons the bond angle decreases from 109 to 1045 H2O Shape The molecular shape of the H2O molecule is bent. Th view the full answer.

H3o Molecular Geometry Shape And Bond Angles Youtube
97 107 ratings The COCl2 molecule has 3 areas of electron repulsion around the central C atom so the shape is trigonal planar.
Lewis structure of the compound h2s shape and bond angles. H2S Lewis Structure The Lewis structure of any compound is a structural representation of the valence electrons participating in the formation of bond along with the nonbonding electron pairs. The Lewis structure of H2S is similar to H2S. In this example we can draw two Lewis structures that are energetically equivalent to each other that is they have the same types of bonds and the same types of formal charges on all of the structuresBoth structures 2 and 3 must be used to represent the molecules structureThe actual molecule is an average of structures 2 and 3 which are called resonance structures.
As the Sulfur atom is less electronegative the overall electronegativity of the compound is less than 04 which makes it nonpolar. This is a review for the advanced placement class. Then identify the LPLP LPBP or BPBP interactions and predict deviations in bond angles.
Lewiss structure of SH2 is really helpful to determine its electron geometry molecular shape number of shared pair and lone pair electrons. Determine the electron group arrangement around the central atom that minimizes repulsions. There are two lone pairs of electrons on the Sulphur atom which makes the geometry of the molecule bent.
This image shows HCN with its constituent atoms the sharing of electrons from one atom to another and the final Lewis Structure showing a single bond to hydrogen and a triple bond to nitrogen. The bond angle is 180 degrees. Knowing the arrangement of atoms distribution of electrons and the shape of the molecule is vastly important in chemistry.
Describe the molecular geometry. Both CCl bonds are polar due to the difference in electronegativity of C and Cl. Molecule Lewis Structure Molecular Shape Bond Angle CS2 Linear 180 CH2O Trigonal Planar 120 H2Se Tetrahedral Bent.
However most of it was discussed druring Honors chemistry. Assign an AX m E n designation. The remaining four electrons are nonbonding electrons and are hence displayed as dots near the Sulfur atom.
H2S has a covalent bond because the sulfur atom completes its octet by sharing 2 electrons with 2 hydrogen atoms and thus forms a covalent bond. SF2 has a simple Lewis structure in which the Sulphur atom is in the centre forming single bonds with both the Fluorine atoms. In fact all of part 1 is basic honors mater.
Molecule of valence e-Lewis Structure NO 3-5 36 1 24e- The Valence Shell Electron Pair Repulsion Theory VSEPR states the electron pairs surrounding an atom tend to repel each other and the shape of the molecule is the result of this electron pair repulsion. To identify and have a complete description of the three-dimensional shape of a molecule we need to know also learn about state the bond angle as well. Then with the Lewis structure we apply the valence-shell electron-pair repulsion VSPER theory to determine the molecular geometry and the electron-group geometry.
Molecular Geometry of Silicon disulfide SiS2 Molecular geometry is a dimensional drawing of the atoms that are forming a molecule. Let us check these fundamentals with the explanation of the lewis structure of H2S. Make a table that contains the Lewis structure molecular shape bond angle and type of hybrid for the following molecules.
As scientists we can predict a lot about how molecules react chemically and their physical properties by looking at Lewis structures and molecular geometry. Now assimilate all the aforementioned steps and draw the Lewis diagram of Silicon disulfide. The bond angles are set at 180.
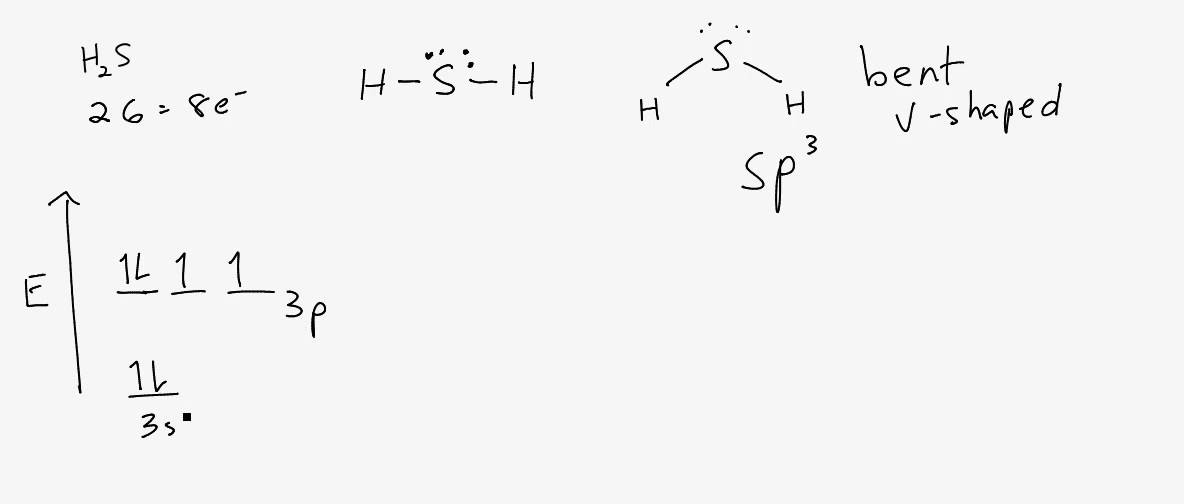
CS2 CH2O H2Se CCl2F2 and NCl3. Through molecular geometry bond length bond type bond angle and other geometrical parameters can be studied. H2S Lewis structure contains two hydrogen and one sulfur atoms.
Sulfur is the central atom and contains 2 lone pairs whereas both hydrogen connected to the central atom with the help of a single bond. Lewis Structures Molecular Geometry Bond Angle etc. Draw the electron dot formula and build the molecular model of nitrate ion NO 3-.
The melting point and boiling point of H2S are -82 and -60 respectively. Draw the Lewis electron structure of the molecule or polyatomic ion. The Sulphur atom has sp3 Hybridization and the bond angle of F-S-F is 98 degrees.

Is H2s Polar Or Nonpolar Dihydrogen Sulfide Youtube

Sf2 Lewis Structure Molecular Geometry Hybridization Polarity And Mo Diagram Techiescientist

H2s Lewis Structure Molecular Geometry Hybridization And Mo Diagram Techiescientist
Lewis Structure Molecular Shape And Hybridization For H2s Youtube
In The Molecule H 2s Four Electron Groups Around The Sulfur Atom Are Arranged In A Tetrahedral Geometry But The Shape Of The Molecule Is Called Bent Why Does The Shape Have A
Pcl3 Molecular Electron Geometry Lewis Structure Bond Angles And Hybridization Geometry Of Molecules
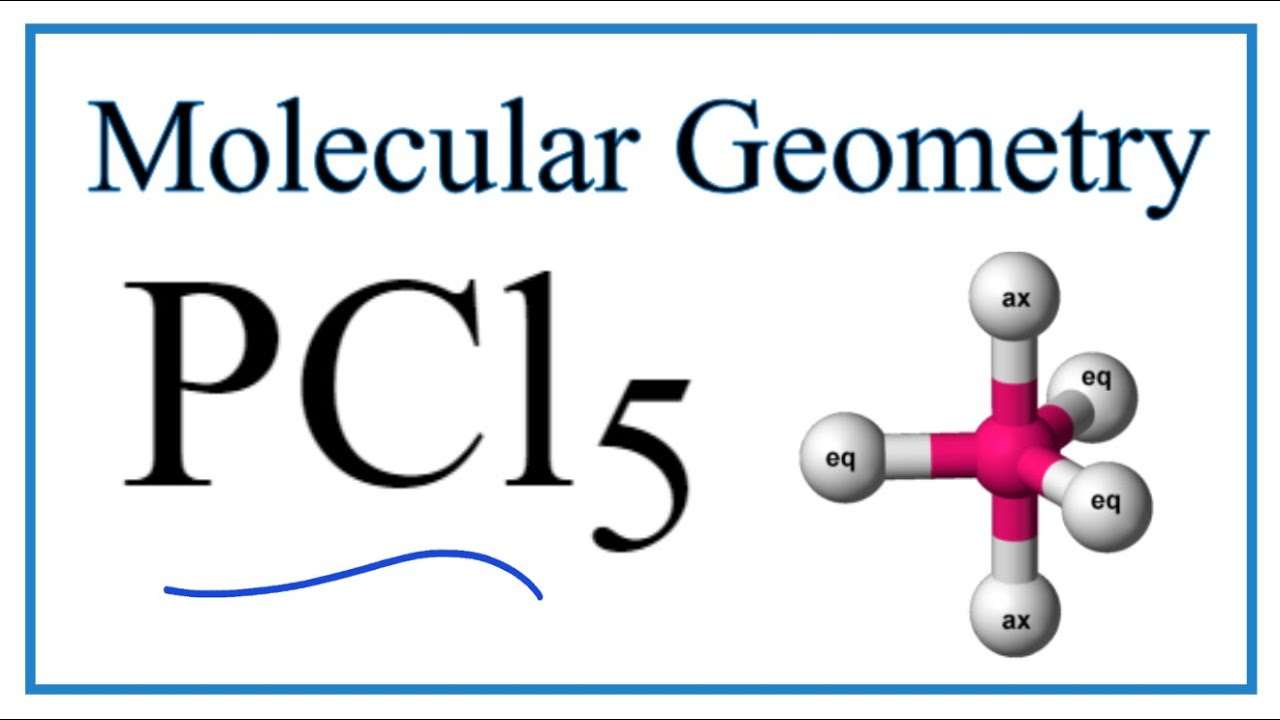
Pcl5 Phosphorous Pentachloride Molecular Geometry Bond Angles Youtube

H2s Lewis Structure Molecular Geometry Hybridization And Mo Diagram Techiescientist

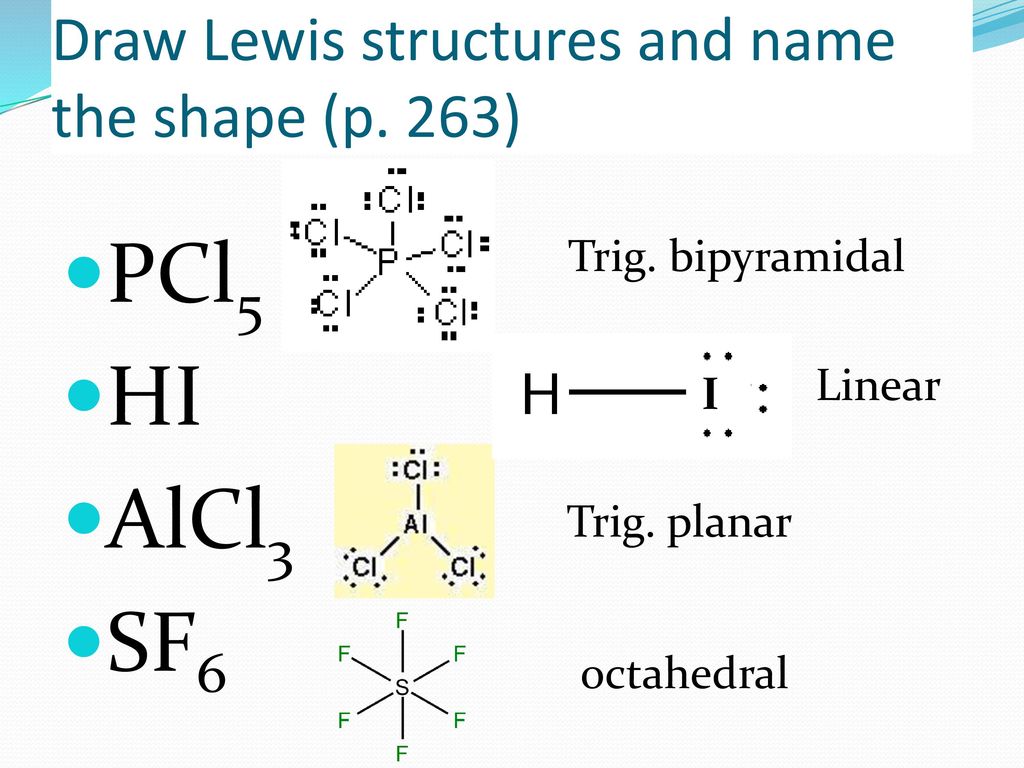
Bell Work P 263 Co2 H2s Draw These Lewis Structures And Name The Shape Ppt Download

Sis2 Lewis Structure Molecular Geometry Hybridization And Polarity Techiescientist

H2s Lewis Structure Molecular Geometry What S Insight

H2s Lewis Structure Molecular Geometry Hybridization And Mo Diagram Techiescientist
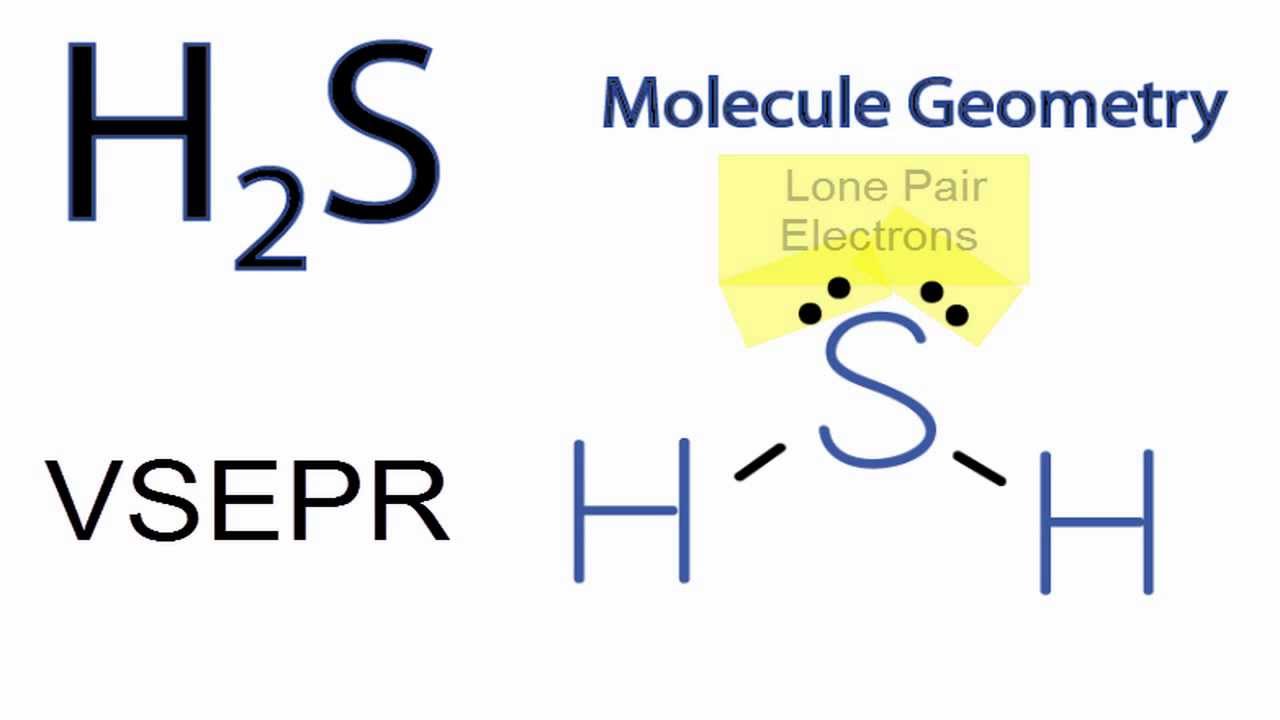
H2s Molecular Geometry Shape And Bond Angles Note Precise Bond Angle Is 92 1 Degrees Youtube
3 7 Geometry And Dipole Moment Chemistry Libretexts
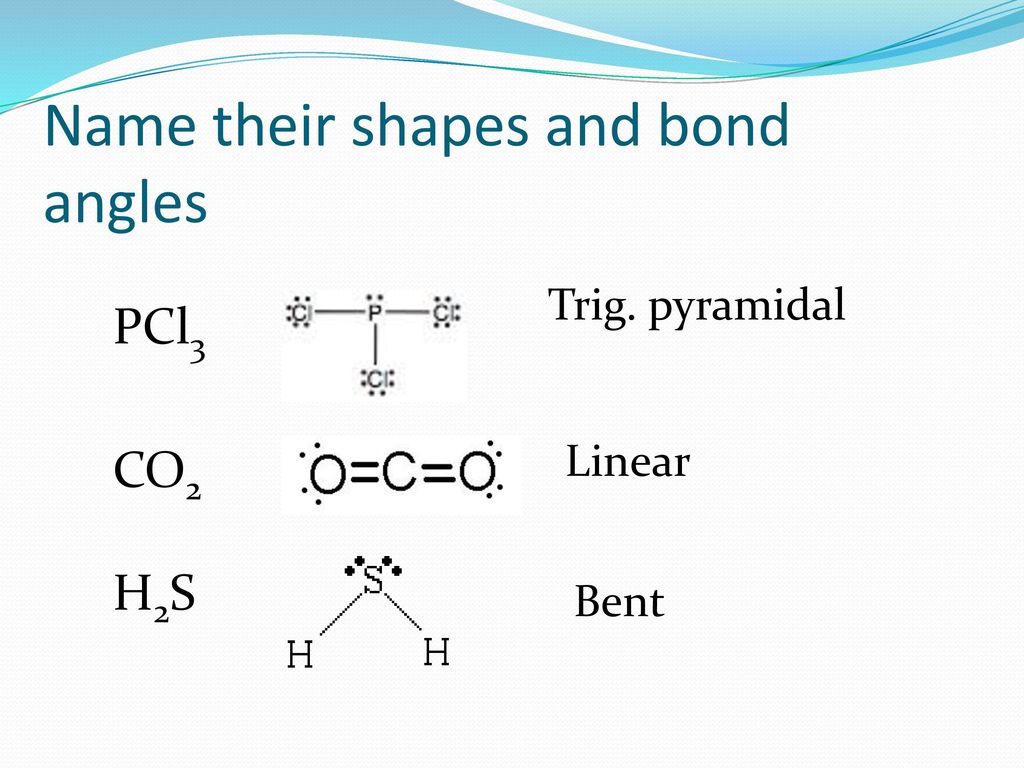
Bell Work P 263 Co2 H2s Draw These Lewis Structures And Name The Shape Ppt Download
Ccl4 Molecular Geometry Lewis Structure Hybridization And Everything
H2s Lewis Structure Molecular Geometry Hybridization And Polarity

Draw The Lewis Structure Of Clbr3 Showing Clutch Prep

H2s Lewis Structure Molecular Geometry What S Insight